|
Molecular mechanisms regulating cell migration
Neural crest cells migrate along conserved paths in the embryo to contribute to numerous traits. How do neural crest cells "know" where to migrate and when to stop? We are interested in understanding the cellular and molecular mechanisms that regulate normal neural crest cell migration. Current work is investigating the role of cell-extracellular matrix interactions and membrane localized proteins in neural crest cell directional movement. This work has implications for understanding cell migration in vivo more generally, such as cancer metastasis. |
|
Expanding the neural crest cell gene regulatory network
Neural crest cells develop from a population of epithelial stem cells at the border of the neural plate. These "neural plate border cells" also give rise to the cranial placodes, and in non-amniotes a unique population of sensory neurons called Rohon-Beard cells. We are interested in understanding how these neural plate border stem cells are segregated into diverse fates including the neural crest. By building on top of a rich history of research into the gene regulatory network that patterns the neural plate border, we are using emerging technologies such as single-cell multiomics and single molecule in situ hybridization to understand stem cell development in vivo. Current work is studying the role of a novel POU domain transcription factor and kruppel-like transcription factors. |
|
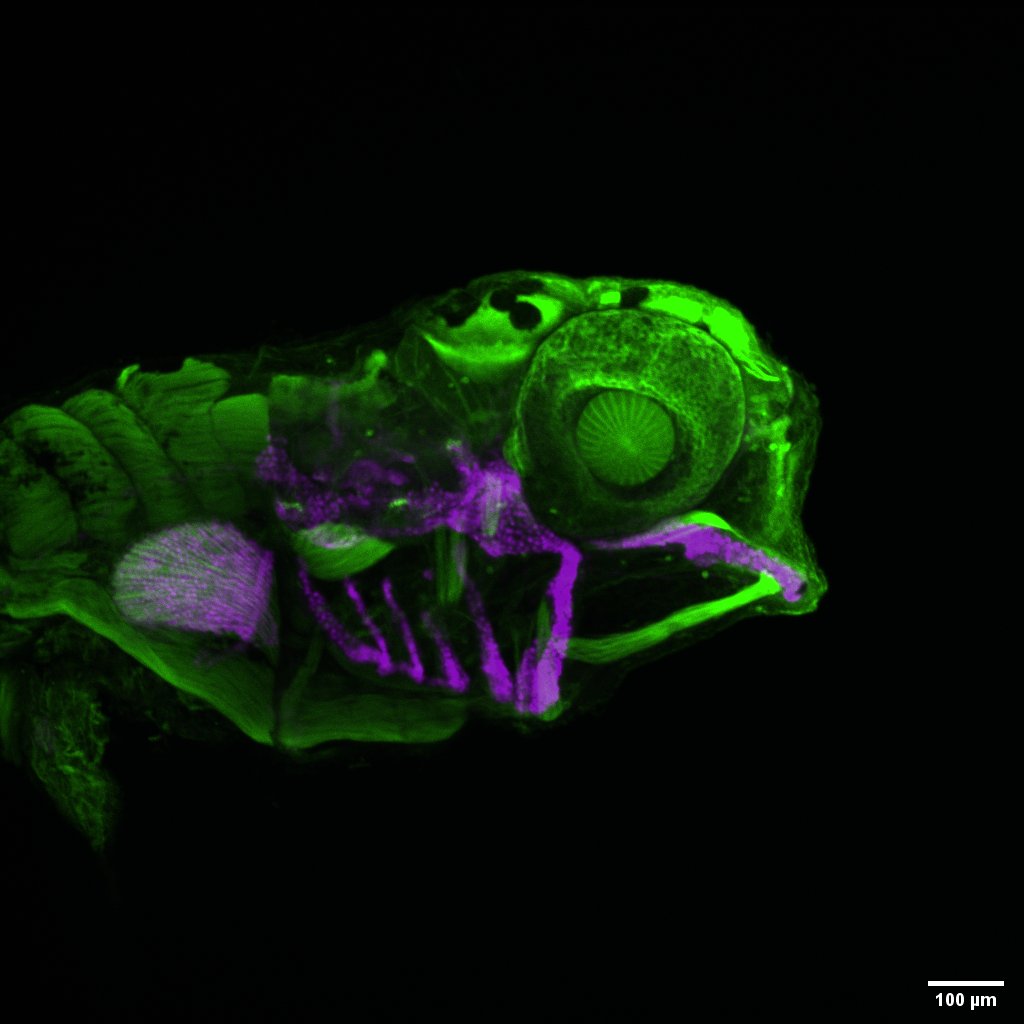
Development of neural crest cell derived traits
Neural crest cells contribute to numerous traits including the craniofacial skeleton, peripheral nervous system, pigment patterns, among others. We are interested in understanding how neural crest cells contribute to these different traits and the gene regulatory interactions underlying morphogenesis of neural crest derived traits. Current projects are investigating the role of hedgehog receptors, cdon and boc, in craniofacial development, and novel POU domain transcription factors in cranial ganglia development. |